Bevacizumab-related pulmonary and renal adverse reactions: A case report
DOI:
https://doi.org/10.5530/ajphs.2024.14.59Keywords:
Bevacizumab, pneumonitis, nephrotic syndromeAbstract
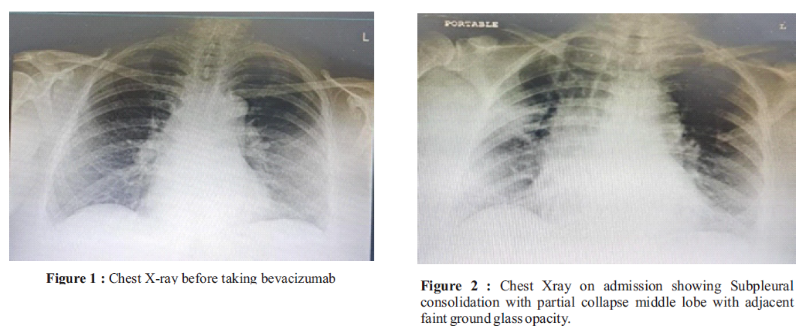
Bevacizumab is a recombinant humanized monoclonal antibody directed against the vascular endothelial growth factor (VEGF). It binds all isoforms of VEGF-A. VEGF is a pro-angiogenic growth factor overexpressed in many solid human cancers, including colorectal cancer. Due to the inhibition of angiogenesis, many adverse reactions such as GI perforations, life-threatening pulmonary bleeding, and increased risk of thromboembolic events have been described. However, its association with pneumonitis and nephrotic syndrome is rarely reported in the literature. We report a case of a 78-year-old woman with stage 4 ovarian cancer who presented with hypertension, pedal edema, nephrotic syndrome, breathlessness, SpO2 fall, and persistent cough after initiation of 3 doses of Bevacizumab. CT scan revealed pneumonitis features, persistent cough, breathlessness on minimal exertion, and dropping SpO2 level. Due to the temporal relationship with the initiation of Bevacizumab, she was suspected to have developed Bevacizumab-induced pneumonitis and nephrotic syndrome. The repeat CT chest showed good response to steroids.